Name:
Natasha Oslinger
Erik Jarvis
9/15/14
Title:
Flame Test Lab
Purpose:
The purpose of this lab was to observe the color and characteristics of certain substances during a flame test.
Procedure:
The methanol handler would pour 5 drops of methanol into the watch glass along with the test substance and combine by gently stirring. We would then light the substance with a match and make observations based on the color and characteristics of the flame.
Pre-lab questions:
What color of light is the lowest in energy?
-Red light is the the lowest energy.
What color of light is the highest in energy?
-Violet light is the highest energy.
What color of light is the highest frequency?
-Violet light is the highest frequency.
What color of light is the lowest frequency?
-Red light has the lowers frequency.
How are electrons “excited”?
-When an electron is in a high energy shell, it tends to be in an excited state.
What does it mean when the electrons are “excited”?
-When electrons are in an excited state, they are of high energy.
In your own words, write a short explanation of how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
-All electrons are in different energy levels, the high the level the more energy the electron has. Electrons can't stay in this high energy state forever, though. When the electron begins to lose its energy, it emits a photon of light. The spacing between when an electron is at a high or low energy will determine where the photon falls on the spectra. In simple terms, what color it will be.
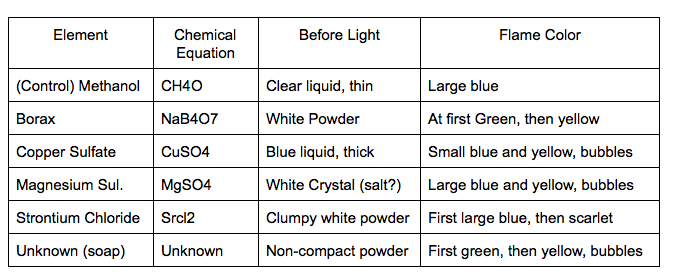
Data Table
Natasha Oslinger
Erik Jarvis
9/15/14
Title:
Flame Test Lab
Purpose:
The purpose of this lab was to observe the color and characteristics of certain substances during a flame test.
Procedure:
The methanol handler would pour 5 drops of methanol into the watch glass along with the test substance and combine by gently stirring. We would then light the substance with a match and make observations based on the color and characteristics of the flame.
Pre-lab questions:
What color of light is the lowest in energy?
-Red light is the the lowest energy.
What color of light is the highest in energy?
-Violet light is the highest energy.
What color of light is the highest frequency?
-Violet light is the highest frequency.
What color of light is the lowest frequency?
-Red light has the lowers frequency.
How are electrons “excited”?
-When an electron is in a high energy shell, it tends to be in an excited state.
What does it mean when the electrons are “excited”?
-When electrons are in an excited state, they are of high energy.
In your own words, write a short explanation of how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
-All electrons are in different energy levels, the high the level the more energy the electron has. Electrons can't stay in this high energy state forever, though. When the electron begins to lose its energy, it emits a photon of light. The spacing between when an electron is at a high or low energy will determine where the photon falls on the spectra. In simple terms, what color it will be.
Data Table
Conclusion:
We were able to observe the different colors of the fames of substances when vaporized during this experiment.
Post Lab Questions:1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
I think the substance has to be heated in order for the electron to be in an excited state releasing the photon of light.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
An ionic bond is normally formed between a non-metal to a metal.
3. What is the definition of a salt in chemistry?
As a result of the neutralization reaction of an acid and a base, an ionic compound known is chemistry as a salt is formed.
4. What is fire?
Fire is the combustion in which a substance is chemically combined with oxygen.
5. What are the chemicals involved in lighting a match?The head of a match is made of sulfur, glass powder, and an oxidizing agent. The strike surface is made of sand, powdered glass, and a chemical called red phosphorus.
6. What is a chemical reaction?
A chemical reactions is the rearrangement of the structure of a substance.
7. What natural cycles does wildfire have an impact on?
Wildfire can impact the carbon cycle, the nitrogen cycle, and hydrologic cycle.
8. List at least three impacts of wildfire in San Diego.
Wildfire can impact the economy due to the cost of firefighting, it can impact the environment by destroying homes and food sources for animals, and can impact the drought by using a lot of water to be put out.
We were able to observe the different colors of the fames of substances when vaporized during this experiment.
Post Lab Questions:1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
I think the substance has to be heated in order for the electron to be in an excited state releasing the photon of light.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
An ionic bond is normally formed between a non-metal to a metal.
3. What is the definition of a salt in chemistry?
As a result of the neutralization reaction of an acid and a base, an ionic compound known is chemistry as a salt is formed.
4. What is fire?
Fire is the combustion in which a substance is chemically combined with oxygen.
5. What are the chemicals involved in lighting a match?The head of a match is made of sulfur, glass powder, and an oxidizing agent. The strike surface is made of sand, powdered glass, and a chemical called red phosphorus.
6. What is a chemical reaction?
A chemical reactions is the rearrangement of the structure of a substance.
7. What natural cycles does wildfire have an impact on?
Wildfire can impact the carbon cycle, the nitrogen cycle, and hydrologic cycle.
8. List at least three impacts of wildfire in San Diego.
Wildfire can impact the economy due to the cost of firefighting, it can impact the environment by destroying homes and food sources for animals, and can impact the drought by using a lot of water to be put out.